Solutions
Asset & Environment Monitoring
Continuous monitoring of critical environments to protect assets, ensure compliance, and maintain stability.
Problem
Critical environments like labs, cleanrooms, and storage facilities require precise monitoring.
Traditional systems are siloed and often lack real‑time alerts or audit trails.
Compliance risks, asset failures, and undetected deviations can lead to downtime, product loss, or regulatory penalties.
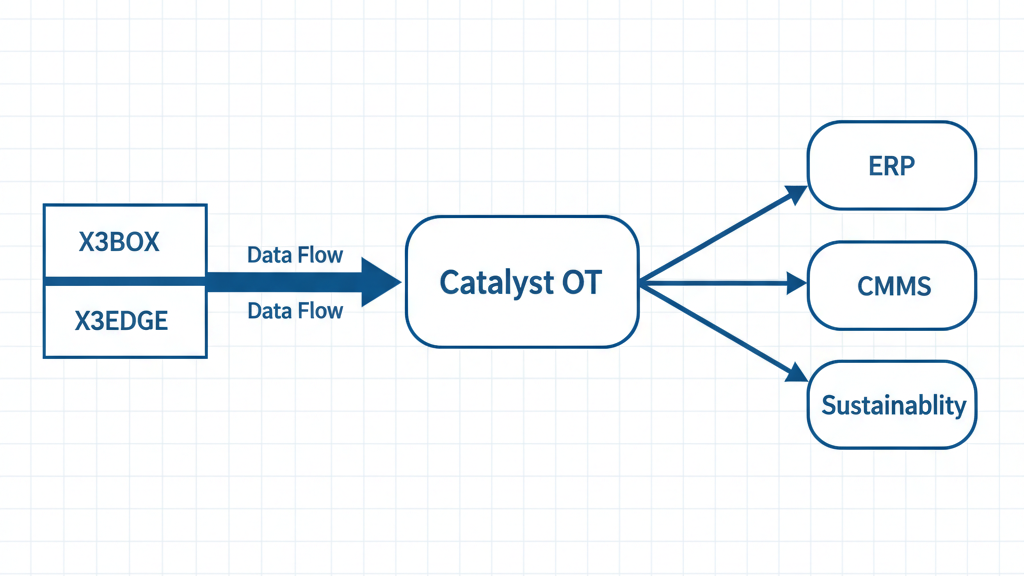
How It Works
1
Edge AI
X3BOX/X3EDGE gather data from sensors (temperature, humidity, vibration, pressure, air quality); buffer and run local models.
2
Platform
Catalyst OT normalizes data, applies thresholds, manages alerts, and provides dashboards/audit logs.
3
Enterprise
ERP/CMMS integration logs deviations, triggers work orders, and supports compliance reporting (FDA, GMP, ISO).
Business Outcomes
A cohesive stack that modernizes plants and networks
Reduced risk of asset or batch loss through early alerts
Stronger compliance with industry standards and audits
Higher uptime and productivity through environment stability
Lower maintenance and insurance costs
Customer Outcomes
What Your Customers Experience
Greater trust from customers who demand compliance and stability
Consistent quality of products and services delivered
Stronger reputation in regulated markets (pharma, healthcare, food)
Key Capabilities
Multi‑sensor monitoring (temperature, humidity, vibration, air quality, pressure)
Compliance dashboards and audit logs
Threshold and anomaly alerts (SMS, email, integrations)
Local dashboards for critical areas; hybrid deployment
Templates for labs, cleanrooms, warehouses, HVAC
Architecture
Edge AI Layer
X3BOX/X3EDGE connect to sensors, buffer locally, run rules/models
Platform Layer
Catalyst OT normalizes, visualizes, logs, and audits
Enterprise Layer
ERP/CMMS integration for compliance, maintenance, and reporting
Rollout Timeline
Site audit, sensor mapping, compliance requirements
Deploy gateways and sensors; dashboards live
Calibrate thresholds/models; set up alerts
Integrate ERP/CMMS; train operators and compliance teams
Case Highlight
Pharma cleanroom: continuous monitoring via X3EDGE sensors reduced deviation incidents by 25%; audit readiness improved significantly.

FAQ
Can the system meet FDA/GMP standards?
Yes—Catalyst OT supports audit trails and compliance dashboards.
What sensors are supported?
Broad library including temperature, humidity, vibration, pressure, air quality.
How are alerts delivered?
Configurable (SMS, email, dashboards, ERP/CMMS).
Can this run fully on‑prem?
Yes—edge and platform can be deployed on‑prem or hybrid.
